Cooperation of Peripheral Hydrogen Atoms for the Stabilization of Aachno-pentaborane (11) with Small Molecules: Hydrogen Bonds and Dihydrogen Bonds
Published 2023-07-01
Keywords
- arachno-pentaborane, dihydrogen bonds, hydrogen bonds, QTAIM, infrared
How to Cite
Abstract
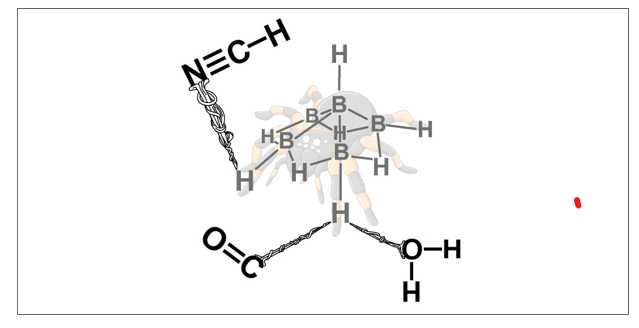
Post-Hartree-Fock calculations performed at the MP2/aug-cc-pVDZ level of theory has been used to analyze the formation of intermolecular complexes between B5H11 and W = CO, NCH, NH3, H2O or HOCH3. The interactions on the structure of the arachno-pentaborane(11) are manifested by the terminal and bridge hydrogen atoms, whereby are formed the hydrogen bonds (H∙∙∙Y with Y = O, C or N) as well as dihydrogen bonds (H∙∙∙H). In this context, the B5H11 shows a host-guest capability for trapping molecules, of course depending on the strength of each aforementioned interactions. The topological descriptors of the Quantum Theory of Atoms in Molecules (QTAIM) were decisive for unveiling each one of the following structures B5H11∙∙∙CO, B5H11∙∙∙NCH, B5H11∙∙∙NH3, B5H11∙∙∙H2O and B5H11∙∙∙HOCH3, and ideally, all hydrogen bonding formed by them.




