Experimental and Kinetic Study of the Effect of using Zr- and Pt-loaded Metals on Y-zeolite-based Catalyst to Improve the Products of n-heptane Hydroisomerization Reactions
Published 2022-10-01
Keywords
- Hydroisomerization process,
- Bimetallic Pt & Zr / Y-zeolite catalysts,
- Reaction kinetics of n-heptane
How to Cite
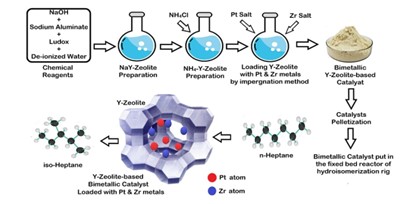
Abstract
The escalating cost of Pt metal has prompted researchers to incorporate other metals into Pt/catalysts to reduce the amount of Pt. In this work, several bimetallic Pt-Zr/HY-zeolite catalysts were prepared by incorporating small amounts of the inexpensive Zr into the Pt/HY-zeolite to form an active and selective catalyst. Results showed that although half of the required platinum metal was used, the catalytic activity of the prepared Pt-Zr/HY bimetallic catalyst was higher than that of the monometallic (Pt or Zr)/HY catalysts, as a result of the improved Lewis acidity of that catalyst that resulted from the addition of the Zr metal; additionally, the branched alkanes' yield also increased. The optimum catalyst was bimetallic, containing 0.5 wt% Pt + 0.5 wt% Zr, which achieved the highest yield of isomers at 70.2 mol%, along with 82.61 and 84.98 mol% for conversion and selectivity, respectively, under 1 MPa and 250°C reaction conditions. In addition, the hydroisomerization reaction kinetic model was achieved, giving good predicted results in agreement with the experimental calculations, with an acceptable relative error. It was found that lower activation energies (about 44.5 kJ/mol) were needed for olefin hydrogenation to iso-paraffins, while higher activation energies were required for i-paraffin hydrocracking (about 138.1kJ/mol).




